The increasing use of mRNA-based applications, both in vials for a speed time to market or in pre-fillable syringes (PFS) for dosing accuracy, poses some technical considerations and challenges during the supply chain and cold storage or during life cycle management.
mRNA solutions being highly sensitive might require low-temperature storage. A PFS system is composed of multiple components and the cold storage will stress the overall system potentially compromising some of the components.
At Stevanato Group, thanks to our Analytical Services team, we investigated the suitability of our containers at freezing conditions focusing on Container Closure Integrity (CCI), functionality and mechanical performance to address the needs of the current and future applications to be at the forefront in supporting the market.
Our EZ-fill® ITC syringes passed all the tests of the main challenges we investigated. Check below the results.
Pre-fillable syringes have several components that compose the system, and deep cold storage is a big challenge to maintaining all the materials' integrity.
We tested and confirmed no loss of CCI of EZ-fill® ITC syringes after 7 days of storage at -70°C. Tests executed through CO2 Headspace Analysis showed no relevant signals of carbon dioxide ingress after a 1-week freezing cycle whilst also verifying the plunger displacement phenomenon confirming the suitability of EZ-fill® ITC system for mRNA applications.
Chart: No CO2 was detected in all 30 ITC filled syringes tested through Carbon Dioxide headspace analysis, indicating that no defect allowing CO2 ingress occurred due to any material property changes at -70°C. The controls in both cases performed as expected, showing a substantial ingress of CO2 for the positive control, and no CO2 ingress for the negative controls.
Low temperature can also affect the functionality of the overall system. Two of the most important parameters that describe the system's functionality are Break Loose and Gliding Force.
We tested - after 1-week storage at -40°C - filled ITC syringes and they showed excellent Break Loose and Gliding Force* performance with comparable results before and after freezing storage.
* ISO 11040-4:2015 as a standard reference
Charts: The test showed that freezing storage has no impact on break loose and gliding force, confirming no functional impact from Room Temperature to -40°C.
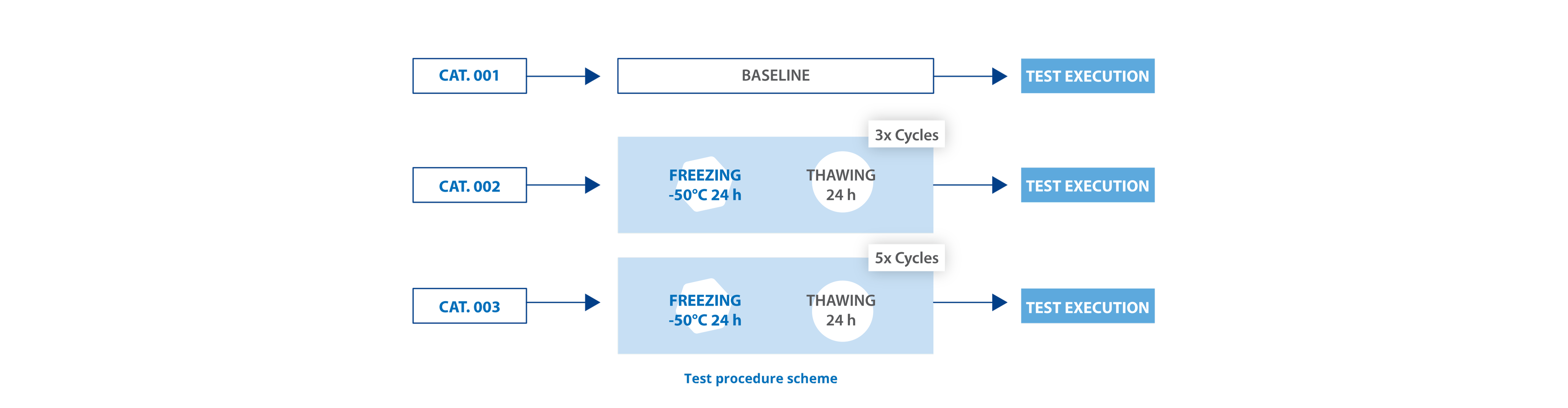
We tested the ITC closure unscrew* after 5 cycles at -50° storage with extraordinary results. The test showed no statistical difference of unscrewing torque (Tmax) between the sub-categories, confirming that ITC closure functionality is guaranteed after freezing condition, with no impact on usability for the final user.
Chart: No evidence of difference of unscrewing force for all the 50 ITC syringes tested for each category.
Deep cold storage may stress the glass barrel of a PFS system causing potential system failures.
Regarding mechanical properties, Burst Test* performed on Stevanato Group ITC syringe did not show difference in glass barrel resistance between -40°C and room temperature storage conditions, confirming good mechanical performances.
*ISO 7458:2004 as standard reference.
Chart: Results at Room Temperature and at -40°C are comparable, confirming that freezing did not affect glass resistance to breakage.
mRNA therapies on the rise: Challenges for primary packaging and solutions to test for best performance