The continued growth of biologics and biosimilars is placing pressure on the performance of primary packaging and the effectiveness of fill-finish operations due to their complex properties.
Managing these typically high-value products means addressing the risk of container breakages, and the risk of particle contamination which is an increasing area of concern for regulatory bodies, as highlighted by the introduction of the revised GMP Annex 1 for the manufacture of sterile medicinal products. In the context of these growing pressures, ready-to-use (RTU) containers are experiencing growing demand.
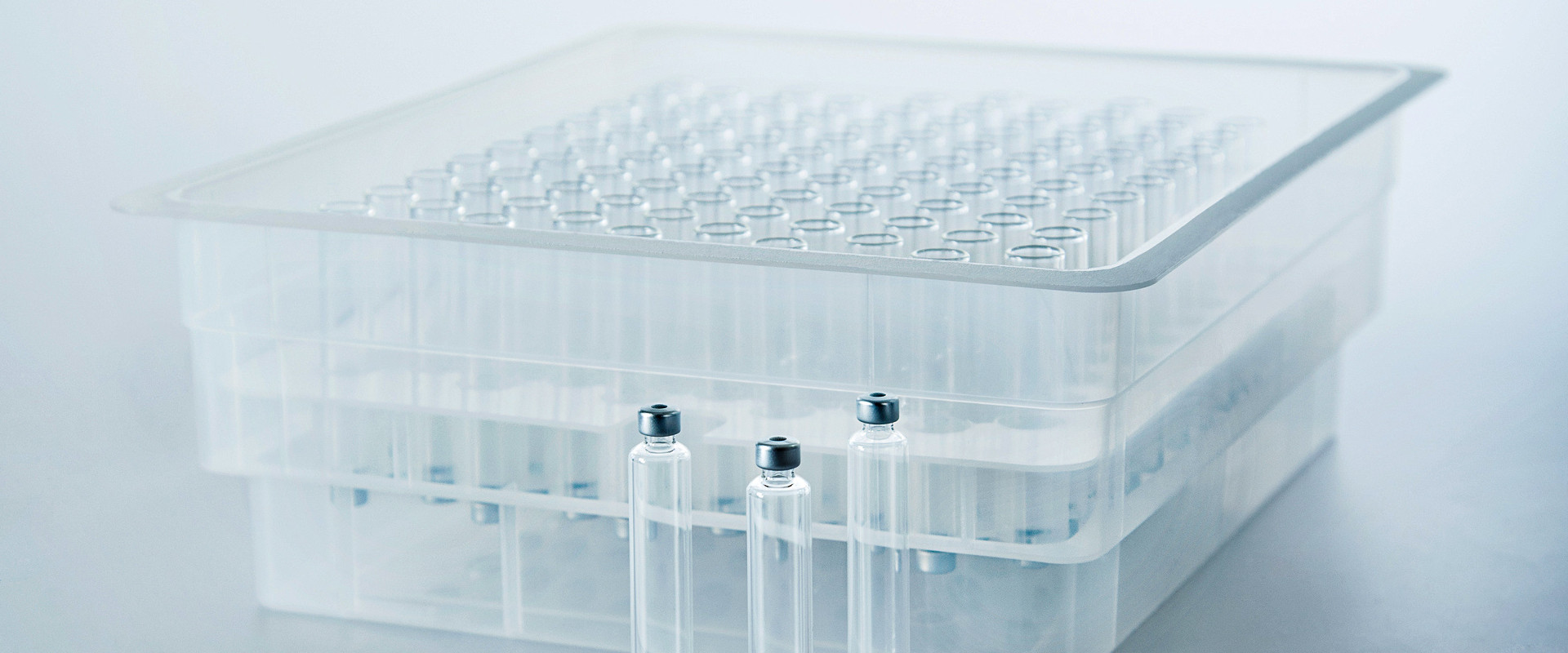
As they are supplied pre-washed, pre-siliconized (if required), and pre-sterilized, these components are ready to be integrated directly into aseptic fill and finish lines, avoiding the need for several preparation procedures to be carried out, including washing, drying, and depyrogenation.
At Stevanato Group, we have extensive experience in fully integrated pre-sterilized containment solutions for aseptic manufacturing and today we celebrate the 10th Anniversary of the EZ-fill® cartridge technology that has become the market leader and the most established platform.
RTU cartridges are used for a broad range of hand-held injection devices such as pens, auto-injectors, and on-body delivery systems, meeting the expectations of increasing numbers of pharma companies that have intensified their focus on drug delivery devices that patients can use in their own homes.
Nexa® EZ-fill® cartridges offer unparalleled quality. Our world-leading process efficiency and operations help reduce the risk of contamination, rejections, complaints and recalls.
Our ready-to-use Nexa® EZ-fill® cartridges are designed for optimizing the fill & finish process and the performance with drug delivery systems offering the following attributes:
- High mechanical resistance and cosmetic quality
- Baked-on siliconization for optimal performance and reduction of particles
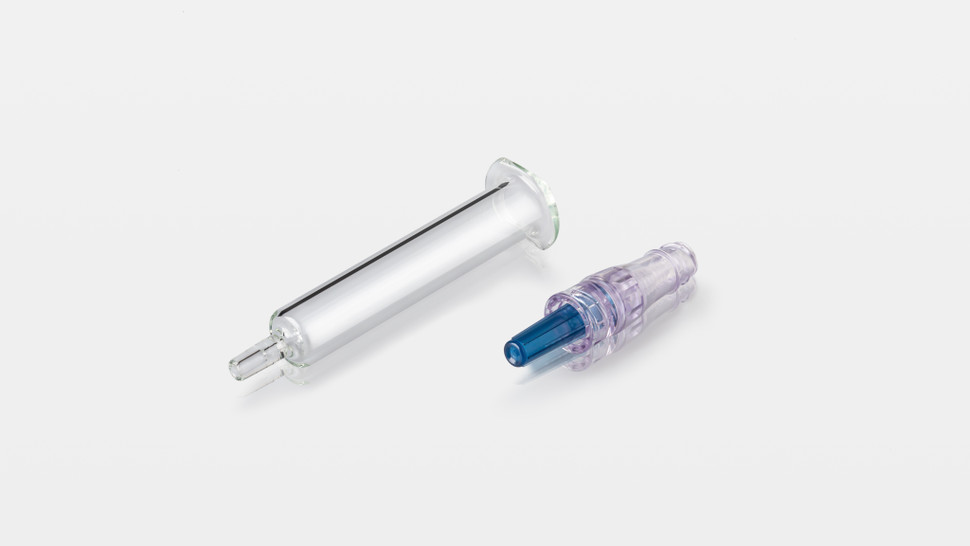
- Widest range of cartridge systems in terms of formats and choice of rubber components
- Crimping robustness based on 100% controls and functional test methods (for pre-capped cartridges)
- No glass-to-glass contact during processing and packaging
- Nest-and-tray configurations, aligned with ISO 21881 and ISO 11040-7
- Single or double steribags for automatic debagging and no-touch transfer technologies
- Tests of drug-container and container-device interaction via Stevanato Group’s Technology Excellence Center
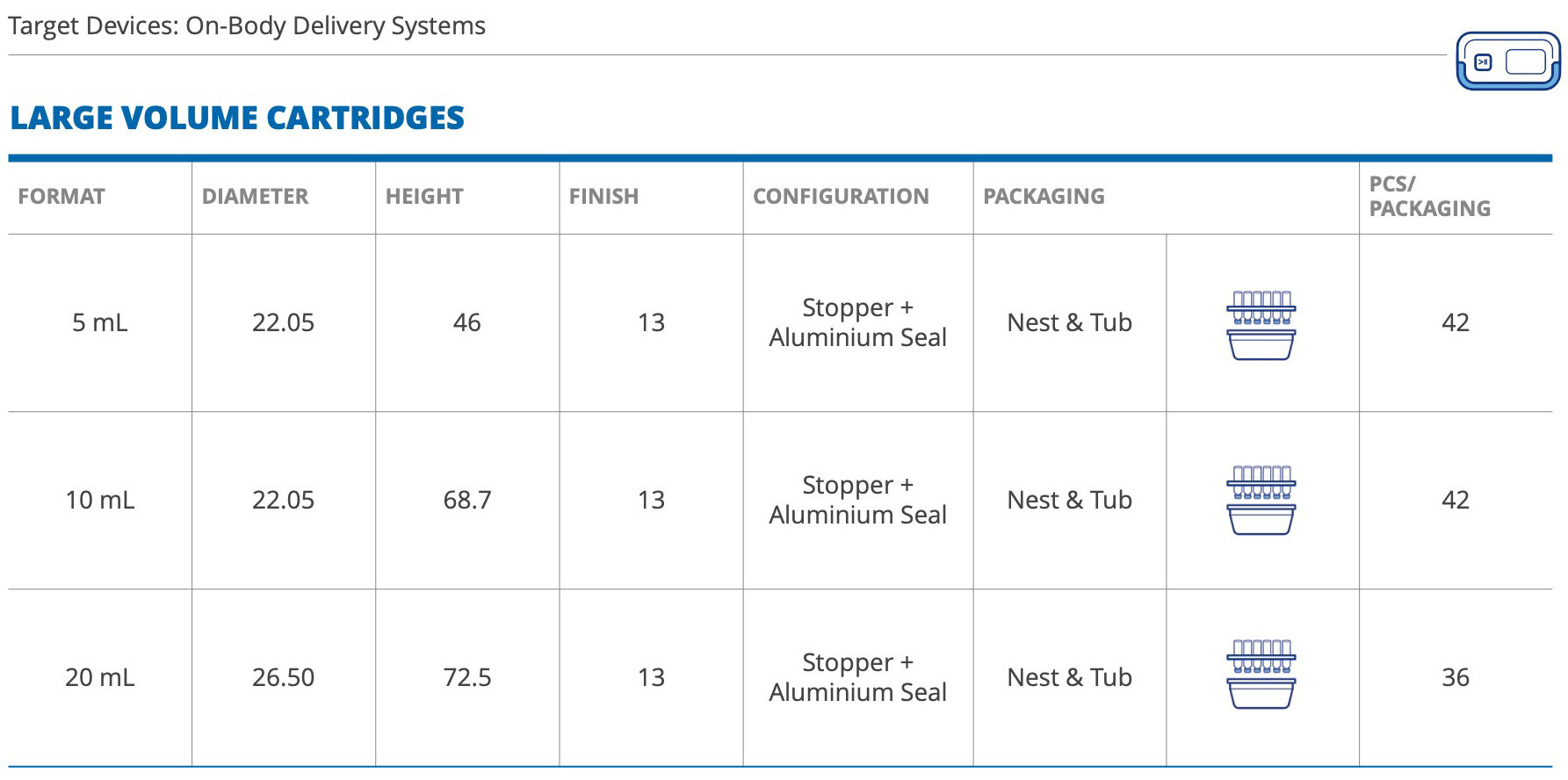
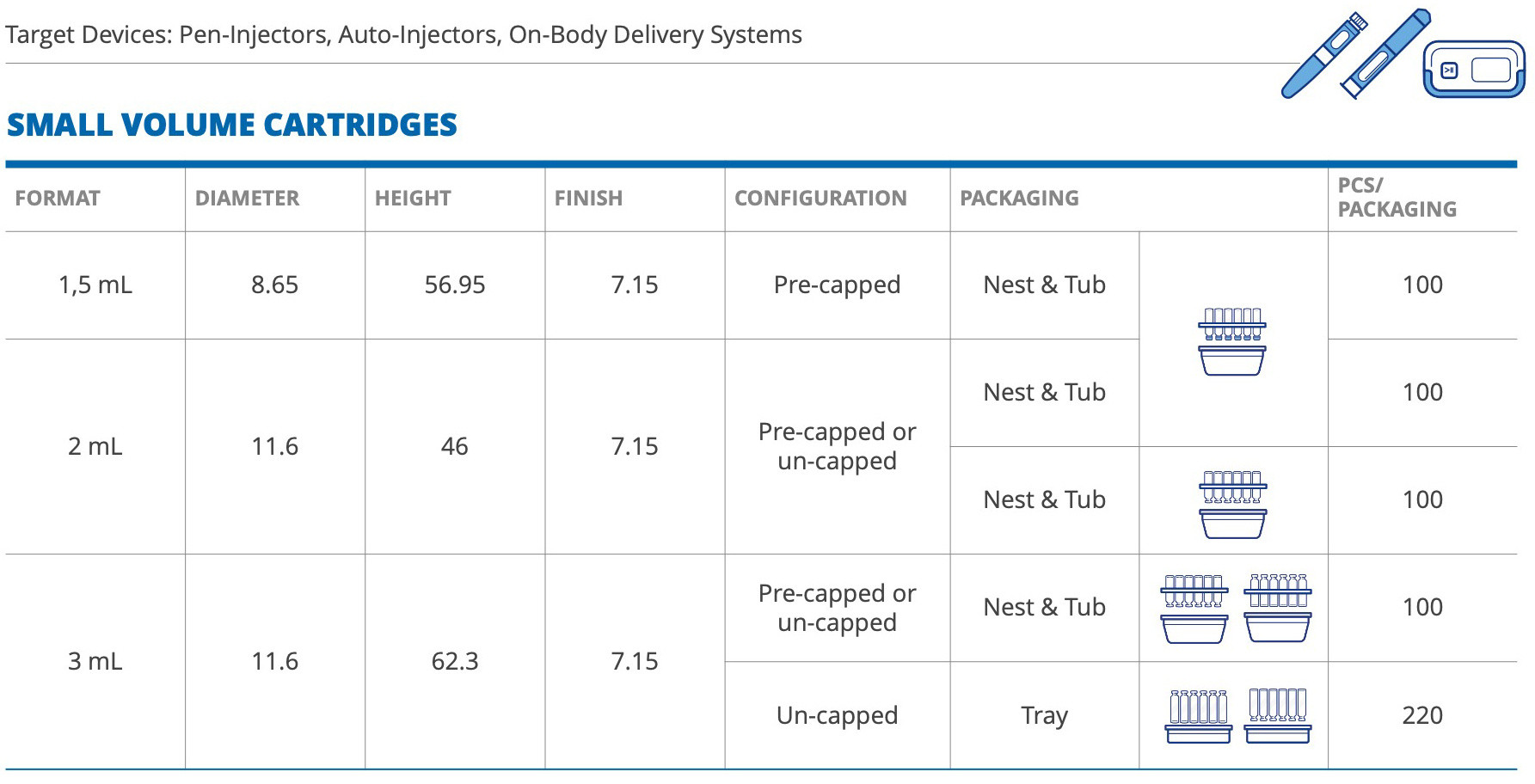
Our portfolio includes both small-volume cartridges – from 1.5mL to 3mL – and large-volume cartridges, from 5mL to 20mL, offering the widest range of validated cartridge formats and related rubber components (combi-seals, front stoppers, and plungers), thus simplifying the choice for pharma companies.
Small-volume RTU cartridges are designed to cope with the demands of delivering precise, small amounts of drugs such as GLP-1 for the treatment of diabetes and obesity, insulin, and hormones – with a choice of nest-and-tub and/or nest-and-tray configurations.
Large-volume RTU cartridges are best suited to the delivery of high-value biologics for the treatment of conditions such as cancer and Parkinson’s and autoimmune diseases.